





Published on Feb 13, 2025
From humans to single-celled bacteria, the life of all organisms depends on the metabolic reactions occuring in their body. Proper functioning of an organism thus depends fundamentally on proper regulation of these biochemical reactions. And it is here that enzymes play a central role. Enzymes are proteins that catalyze (i.e., increaseor decrease the rates of)chemical reactions. In enzymatic reactions, the molecules at the beginning of the process are called substrates, and they are converted into different molecules, called the products. Almost all processes in a biological cell need enzymes to occur at significant rates. Since enzymes are selective for their substrates and speed up only a few reactions from among many possibilities, the set of enzymes made in a cell determines which metabolic pathways occur in that cell.
Like all catalysts, enzymes work by lowering the activation energy (Ea‡) for a reaction, thus dramatically increasing the rate of the reaction. As a result, products are formed faster and reactions reach their equilibrium state more rapidly. Most enzyme reaction rates are millions of times faster than those of comparable un-catalyzed reactions. As with all catalysts, enzymes are not consumed by the reactions they catalyze.
Enzyme activity can be affected by other molecules. Inhibitors are molecules that decrease enzyme activity; activators are molecules that increase activity. Many drugs and poisons are enzyme inhibitors. Activity is also affected by temperature, chemical environment (e.g., pH), and the concentration of substrate. Some enzymes are used commercially, for example, in the synthesis of antibiotics.
Ribbon diagram showing human carbonic anhydrase II. The grey sphere is the zinc cofactor in the active site. Diagram drawn from PDB 1MOO. Enzymes are generally globular proteins and range from just 62 amino acid residues in size to over 2,500 residues. A small number of RNA-based biological catalysts exist, with the most common being the ribosome; these are referred to as either RNA-enzymes or ribozymes. The activities of enzymes are determined by their three-dimensional structure. However, although structure does determine function, predicting a novel enzyme's activity just from its structure is a very difficult problem that has not yet been solved.
Most enzymes are much larger than the substrates they act on, and only a small portion of the enzyme (around 3–4 amino acids) is directly involved in catalysis. The region that contains these catalytic residues, binds the substrate, and then carries out the reaction is known as the active site. Enzymes can also contain sites that bind cofactors, which are needed for catalysis. Some enzymes also have binding sites for small molecules, which are often direct or indirect products or substrates of the reaction catalyzed. This binding can serve to increase or decrease the enzyme's activity, providing a means for feedback regulation.
Like all proteins, enzymes are long, linear chains of amino acids that fold to produce a three-dimensional product. Each unique amino acid sequence produces a specific structure, which has unique properties. Individual protein chains may sometimes group together to form a protein complex. Most enzymes can be denatured—that is, unfolded and inactivated—by heating or chemical denaturants, which disrupt the three-dimensional structure of the protein. Depending on the enzyme, denaturation may be reversible or irreversible.
Enzymes are usually very specific as to which reactions they catalyze and the substrates that are involved in these reactions. Complementary shape, charge and hydrophilic/hydrophobic characteristics of enzymes and substrates are responsible for this specificity.
Some of the enzymes showing the highest specificity and accuracy are involved in the copying and expression of the genome. These enzymes have "proof-reading" mechanisms. Here, an enzyme such as DNA polymerase catalyzes a reaction in a first step and then checks that the product is correct in a second step. This two-step process results in average error rates of less than 1 error in 100 million reactions in high-fidelity mammalian polymerases. Similar proofreading mechanisms are also found in RNA polymerase, aminoacyl tRNA synthetases and ribosomes.
Enzymes are very specific, and it was suggested by the Nobel laureate organic chemist Emil Fischer in 1894 that this was because both the enzyme and the substrate possess specific complementary geometric shapes that fit exactly into one another. This is often referred to as "the lock and key" model. However, while this model explains enzyme specificity, it fails to explain the stabilization of the transition state that enzymes achieve.
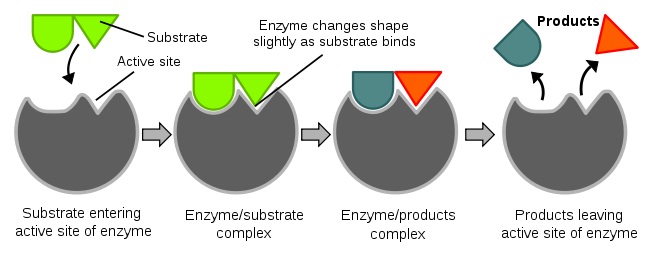
Diagrams to show the induced fit hypothesis of enzyme action.
In 1958, Daniel Koshland suggested a modification to the lock and key model: since enzymes are rather flexible structures, the active site is continually reshaped by interactions with the substrate as the substrate interacts with the enzyme. In some cases, such as glycosidases, the substrate molecule also changes shape slightly as it enters the active site. The active site continues to change until the substrate is completely bound, at which point the final shape and charge is determined.
Enzymes can act in several ways, all of which lower ΔG+:
Lowering the activation energy by creating an environment in which the transition state is stabilized (e.g. straining the shape of a substrate—by binding the transition-state conformation of the substrate/product molecules, the enzyme distorts the bound substrate(s) into their transition state form, thereby reducing the amount of energy required to complete the transition).
Lowering the energy of the transition state, but without distorting the substrate, by creating an environment with the opposite charge distribution to that of the transition state.
Providing an alternative pathway. For example, temporarily reacting with the substrate to form an intermediate ES complex, which would be impossible in the absence of the enzyme.
Reducing the reaction entropy change by bringing substrates together in the correct orientation to react. Considering ΔH‡ alone overlooks this effect.
The internal dynamics of enzymes is linked to their mechanism of catalysis. Internal dynamics are the movement of parts of the enzyme's structure, such as individual amino acid residues, a group of amino acids, or even an entire protein domain. These movements occur at various time-scales ranging from femtoseconds to seconds. Networks of protein residues throughout an enzyme's structure can contribute to catalysis through dynamic motions. However, although these movements are important in binding and releasing substrates and products, it is not clear if protein movements help to accelerate the chemical steps in enzymatic reactions. These new insights also have implications in understanding allosteric effects and developing new drugs.
Allosteric transition of an enzyme between R and T states, stabilized by an agonist, an inhibitor and a substrate (the MWC model)
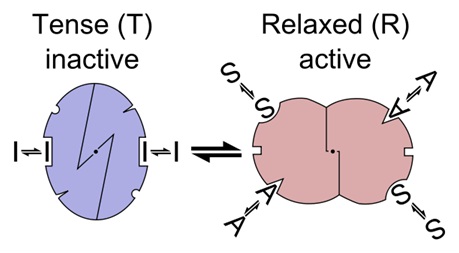
Allosteric sites are sites on the enzyme that bind to molecules in the cellular environment. The sites form weak, noncovalent bonds with these molecules, causing a change in the conformation of the enzyme. This change in conformation translates to the active site, which then affects the reaction rate of the enzyme. Allosteric interactions can both inhibit and activate enzymes and are a common way that enzymes are controlled in the body.
Some enzymes do not need any additional components to show full activity. However, others require non-protein molecules called cofactors to be bound for activity. Cofactors can be either inorganic (e.g., metal ions and iron-sulfur clusters) or organic compounds. Organic cofactors can be either prosthetic groups, which are tightly bound to an enzyme, or coenzymes, which are released from the enzyme's active site during the reaction. Coenzymes include NADH, NADPH and adenosine triphosphate. These molecules transfer chemical groups between enzymes.
Space-filling model of the coenzyme NADH
Coenzymes are small organic molecules that can be loosely or tightly bound to an enzyme. Tightly bound coenzymes can be called allosteric groups. Coenzymes transport chemical groups from one enzyme to another. The chemical groups carried include the hydride ion (H-) carried by NAD or NADP+, the phosphate group carried by adenosine triphosphate, the acetyl group carried by coenzyme A, formyl, methenyl or methyl groups carried by folic acid and the methyl group carried by S-adenosylmethionine.
Since coenzymes are chemically changed as a consequence of enzyme action, it is useful to consider coenzymes to be a special class of substrates, or second substrates, which are common to many different enzymes. For example, about 700 enzymes are known to use the coenzyme NADH.
Coenzymes are usually continuously regenerated and their concentrations maintained at a steady level inside the cell: for example, NADPH is regenerated through the pentose phosphate pathway. This continuous regeneration means that even small amounts of coenzymes are used very intensively. For example, the human body turns over its own weight in ATP each day.
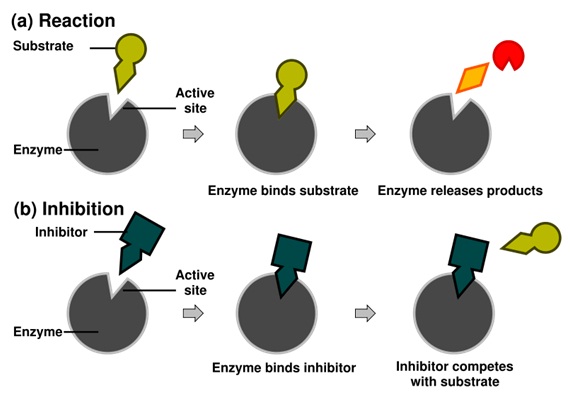
Competitive inhibitors bind reversibly to the enzyme, preventing the binding of substrate. On the other hand, binding of substrate prevents binding of the inhibitor. Substrate and inhibitor compete for the enzyme.
The coenzyme folic acid (left) and the anti-cancer drug methotrexate (right) are very similar in structure. As a result, methotrexate is a competitive inhibitor of many enzymes that use folates.
In competitive inhibition, the inhibitor and substrate compete for the enzyme (i.e., they can not bind at the same time). Often competitive inhibitors strongly resemble the real substrate of the enzyme. For example, methotrexate is a competitive inhibitor of the enzyme dihydrofolate reductase, which catalyzes the reduction of dihydrofolate to tetrahydrofolate. The similarity between the structures of folic acid and this drug are shown in the figure.
In uncompetitive inhibition the inhibitor can not bind to the free enzyme, but only to the ES-complex. The EIS-complex thus formed is enzymatically inactive. This type of inhibition is rare, but may occur in multimeric enzymes.
Non-competitive inhibitors can bind to the enzyme at the binding site at the same time as the substrate,but not to the active site. Both the EI and EIS complexes are enzymatically inactive. Because the inhibitor can not be driven from the enzyme by higher substrate concentration (in contrast to competitive inhibition), the apparent Vmax changes. But because the substrate can still bind to the enzyme, the Km stays the same.
This type of inhibition resembles the non-competitive, except that the EIS-complex has residual enzymatic activity. This type of inhibitor does not follow Michaelis-Menten equation.
In many organisms inhibitors may act as part of a feedback mechanism. If an enzyme produces too much of one substance in the organism, that substance may act as an inhibitor for the enzyme at the beginning of the pathway that produces it, causing production of the substance to slow down or stop when there is sufficient amount. This is a form of negative feedback. Enzymes which are subject to this form of regulation are often multimeric and have allosteric binding sites for regulatory substances. Their substrate/velocity plots are not hyperbolar, but sigmoidal (S-shaped).
Irreversible inhibitors react with the enzyme and form a covalent adduct with the protein. The inactivation is irreversible. These compounds include eflornithine a drug used to treat the parasitic disease sleeping sickness. Penicillin and Aspirin also act in this manner. With these drugs, the compound is bound in the active site and the enzyme then converts the inhibitor into an activated form that reacts irreversibly with one or more amino acid residues.
Enzymes serve a wide variety of functions inside living organisms. They are indispensable for signal transduction and cell regulation, often via kinases and phosphatases. They also generate movement, with myosin hydrolysing ATP to generate muscle contraction and also moving cargo around the cell as part of the cytoskeleton. Other ATPases in the cell membrane are ion pumps involved in active transport. Enzymes are also involved in more exotic functions, such as luciferase generating light in fireflies. Viruses can also contain enzymes for infecting cells, such as the HIV integrase and reverse transcriptase, or for viral release from cells, like the influenza virus neuraminidase.
An important function of enzymes is in the digestive systems of animals. Enzymes such as amylases and proteases break down large molecules (starch or proteins, respectively) into smaller ones, so they can be absorbed by the intestines. Starch molecules, for example, are too large to be absorbed from the intestine, but enzymes hydrolyse the starch chains into smaller molecules such as maltose and eventually glucose, which can then be absorbed. Different enzymes digest different food substances. In ruminants which have herbivorous diets, microorganisms in the gut produce another enzyme, cellulase to break down the cellulose cell walls of plant fiber.
Several enzymes can work together in a specific order, creating metabolic pathways. In a metabolic pathway, one enzyme takes the product of another enzyme as a substrate. After the catalytic reaction, the product is then passed on to another enzyme. Sometimes more than one enzyme can catalyze the same reaction in parallel, this can allow more complex regulation: with for example a low constant activity being provided by one enzyme but an inducible high activity from a second enzyme.
Glycolytic enzymes and their functions in the metabolic pathway of glycolysis
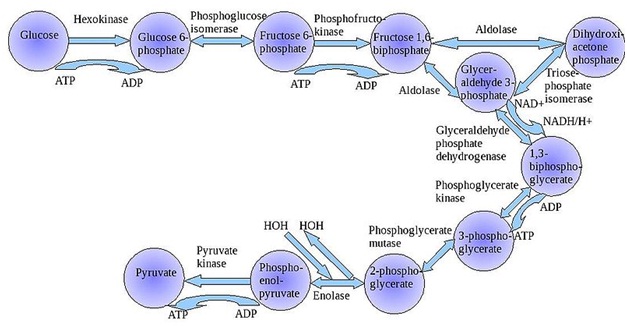
Enzymes determine what steps occur in these pathways. Without enzymes, metabolism would neither progress through the same steps, nor be fast enough to serve the needs of the cell. Indeed, a metabolic pathway such as glycolysis could not exist independently of enzymes. Glucose, for example, can react directly with ATP to become phosphorylated at one or more of its carbons. In the absence of enzymes, this occurs so slowly as to be insignificant. However, if hexokinase is added, these slow reactions continue to take place except that phosphorylation at carbon 6 occurs so rapidly that if the mixture is tested a short time later, glucose-6-phosphate is found to be the only significant product. Consequently, the network of metabolic pathways within each cell depends on the set of functional enzymes that are present.
Enzymes are used in the chemical industry and other industrial applications when extremely specific catalysts are required. However, enzymes in general are limited in the number of reactions they have evolved to catalyze and also by their lack of stability in organic solvents and at high temperatures. Consequently, protein engineering is an active area of research and involves attempts to create new enzymes with novel properties, either through rational design or in vitro evolution. These efforts have begun to be successful, and a few enzymes have now been designed "from scratch" to catalyze reactions that do not occur in nature.
1. Comprehensive Laboratory Manual In Biology-XII 2. Biology Text For Class XII – NCERT
2. http://www.wikipedia.org/