





Published on Feb 13, 2025
• Urine is a typically sterile liquid by-product of the body secreted by the kidneys.
• Major composition of urine is water (97%). Other component of urine are organic & inorganic compounds like urea,bilirubin, ammonium chloride and salts of calcium & potassium .Also they excrete excess hormones & vitamins
• It is the series of test done on urine. It is the one of the most common method of medical diagnostic.
Most of the blood constituents are present in urine. Most of the body wastes are brought to kidney for filtration. Hence the body ailment can be located through this analysis.
Most of the bacteria and viruses are discharged from our body through urine.
This method is more convenient than blood test because:
Some might be scared of blood so this provides to be an alternative for blood tests.
It is easy to analyze most of the bacteria in water medium than in blood.
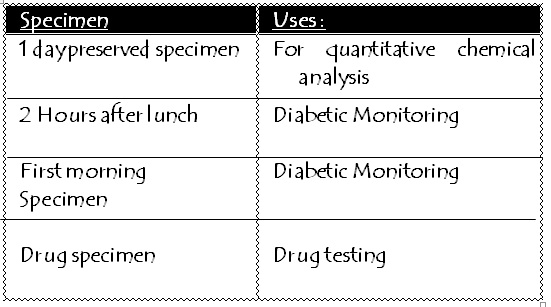
Urine is stored in the refrigerator which doesn’t affect the constituent of urine.
Required Volume:30-45ml taken within 4 minutes
Temperature:32.5ºC to37.7ºC
This process is done under following reagents :-
Phenol: causesanodor change
Toluene: noteffectivefor bacteriaand molds
Thymol crystals:preservesglucoseand sedimentswell
Formalin:excellent sediment preservative
Boricacid: preserves proteinwell
o Colour :Denotes the degree of concentration. The color of non-pathological urine varies widely from colorless to deep yellow; the more concentrated the urine, the deeper the color. The color of urine is usually described after visual inspection with common color terms.
o PH:Denotes the presence of H+ ion concentration in the content sample.
Examination : Using PH papers .
Urine sample
Benedict’s solution
Fehling’s solution – 1
Fehling’s solution – 2
Copper sulphate solution
Sodium hydroxide
Borsch’s reagent
Ethanol
Test tubes
Used to test amount of albumin present in the given content sample . Examination :Done using sodium hydroxide & copper sulphate . This forms thick blue ppt.
Used to test the presence of phenol based compounds present in urine . Examination:Mixing the given urine soln. with neutral FeCl3. A light violet colouring is observed .
This test is used to detect the type of amino group that is excreted through urine . These amines may be primary (1), secondary(2), tertiary(3) .
i) The given sample of urine is taken in a test tube .To the soln. alcoholic KOH is added along with CHCl3 . A foul smelling gas poisonous gas is released (R-CN) . This indicates that the given urine has the presence of 1amine . If there is no smell , then 1amine is absent .
o Carbonyl group test :This test is used to determine the presence of carbonyl group namely KETONES(-CO-) & ALDEHYDES (-CHO) .
I. Urine sample is mixed with BORSCH’s reagent , in presence of alcohol it , there forms a yellow ppt. which confirms the presence of carbonyl group .
II. FEHLING TEST : This test is employed to test the presence of reducible groups .
The sample is taken and FEHLING – A(CuSO4) & FEHLING - B (Sodium Potassium Tartrate ) are added and water bathed . Presence of red ppt. (Cu2O)confirms the presence of carbonyl group .
i. TOULENETEST:Given sample is mixed with TOLUENE’s reagent (Ammo. AgNO3) & heated . If The sample doesn’t show any kind of silver mirror then ALDEHYDE is absent
ii. K2Cr2O7TEST :Given urine is mixed with K2Cr2O7and water bathed . If the soln. imparts green colour (CrO3 ion). Then it confirms that there is ALDEHYDE present in the urine sample .
iii. KETONE TEST:The sample urine is mixed with alcohol and SCHIFF’s Base it forms violet colouration . This confirms the presence of Ketone
Sample urine is taken in a test tube and BENEDICT’s solutions is added .This mixture is kept in water bath , after some time it produces colouration depending upon the concentration of glucose .
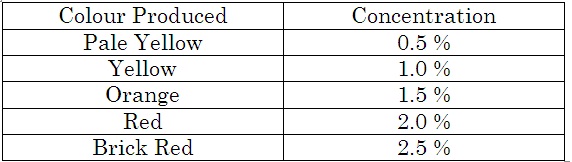
In the given sample of urine specimen, the values of PH , Blood Glucose level and Protein are the serious one . Higher albumin (protein) & PH level in urine indicates serious trouble in Kidney. The person is also affected by Diabetes
• Class XI practical textbook .
• http://en.wikipedia.org/wiki/Benedict%27s_reagent
• http://www.chemguide.co.uk/organicprops/carbonyls/oxidation.html
• http://www.texascollaborative.org/spencer_urinalysis/ds_sub2.htm
• http://www.austincc.edu/mlt/ua/ua_links.html