





Published on Feb 13, 2025
Milk is considered an idea food as it contains most of the constituents of our diet. It is a rich source of proteins and fats. It also contains calcium and phosphorous in sufficient quantity. Therefore, milk is considered to be an important diet for the growing children, and pregnant and lactating women. The milk of buffalo, cow and goat are commonly used for human consumption. Milk is slightly heavier than water (specific gravity = 1.035) and has a slightly acidic pH (pH =6.7).

The various constituents of milk are
1. Water - 87.3%
2. Butter fat - 3.8%
3. Casein (a protein) - 2.5%
4. Albumin and globulin - 0.7%
(Other proteins)
5. Lactose (milk sugar) - 5.0%
6. Ash (minerals) - 0.7%
Casein is the principal type of protein present in the milk. It represents about 80% protein nitrogen of the milk. B-lacto globulins and a-lactalbumin are the other proteins of the milk. Some antibodies called immunoglobulin present in milk are also proteins.
Casein can be coagulated by acid, rennet and heating. It is a coaguable protein. The other milk proteins i.e. b-lactoglubind and a-lacto albumins are called non-coaguable proteins as they can be coagulated only by heating.
The study of presence of coaguable and non-coaguable proteins in milk has been taken in this project.
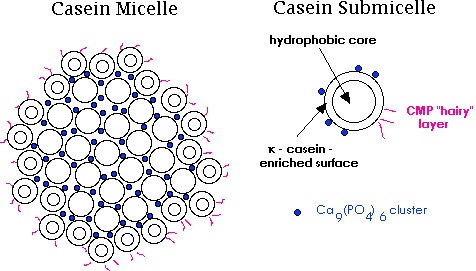
Casein is the name for a family of related phosphoprotein proteins. These Proteins are commonly found in mammalian milk, making up 80% of the proteins in cow milk and between 60% and 65% of the proteins in human milk. Casein has a wide variety of uses, from being a major component of cheese, to use as a food additive, to a binder for safety matches. As a food source casein supplies amino acids; carbohydrates; and two inorganic elements, calcium and phosphorus.
Is a mixture of proteins isolated from whey, the liquid material created by-product of cheese production. Some preclinical studies in rodents have suggested that whey protein may possess anti-inflammatory or anti-cancer properties; however, human data is lacking. The effects of whey protein on human health are of great interest and are currently being investigated as a way of reducing disease risk, as well as a possible supplementary treatment for several diseases. Whey protein is commonly marketed and ingested as a dietary and various health claims have been attributed to it in the alternative community. Although whey proteins are responsible for some milk the major allergens in milk are the caseins.
The thermal coagulation of unfractionated whey proteins was inhibited by various sugars. The disaccharides, sucrose and lactose, were most effective, and the amino sugar, glucosamine, least effective in this respect. Ultraviolet absorption and light-scattering measurements on the thermal denaturation and coagulation of both unfractionated and individual whey proteins (b-lactalbumin, b-lacto globulin, and bovine serum albumin) showed that sucrose promotes the denaturation of these proteins but inhibits their subsequent coagulation. These results are interpreted in terms of the effect of sucrose on the hydrophobic interactions between solvent and protein.
1. Milk
2. Test tubes
3. Rennin tablets
4. Millon’s reagent
5. Beaker
6. Burner

1. Take about 20ml of milk in a beaker. Grind a rennin tablet and add it to the milk.
2. Warm the milk. The milk will coagulate.
3. Filter the coagulated (solidified) milk. The liquid filtrate is whey.
4. Take a small amount of whey in separate test tubes and test it for the presence of protein by using Millon’s reagent.
Whey gives positive test for protein, indicating the presence of non coaguable proteins in whey.

Cow and buffalo milks contains both coaguable and non-coaguable proteins.
• Comprehensive Laboratory Manual in Biology
• Wikipedia