





Published on Feb 13, 2025
The objective: I want to determine which carbon nanostructure is the strongest: carbon arranged in sheets (graphite), carbon arranged in hollow spheres (fullerenes), or carbon packed together closely (diamond). Using these tiny nanostructures helps scientists create a new range of materials that are lighter and stronger than ever before.
In nature, carbon can bond to itself to form super strong sheets like those in graphite. A single sheet of graphite is very stable, strong, and flexible. The problem is it does not bind very strongly to other sheets. The carbon structures arranged in sheets can move easily, making them flaky and easily breakable, like the graphite centre of pencils. Using nanotechnology, scientists can roll or fold these carbon sheets in the laboratory into three-dimensional structures, creating different fullerenes like nanotubes. These carbon nanostructures have amazing properties.
The purpose of my project is to find out which carbon nanostructure is the strongest: six stacked sheets representing graphite, rolls representing fullerenes, and a cube representing diamond
I think that the carbon nanostructure, diamond will be the strongest.
1. Plastic disposable cup
2. 1-hole puncher
3. Curling ribbon (24 inches long)
4. Ruler
5. Scissors
6. Oragami paper, 7 x 7-inch square (60 sheets)
7. Tape
8. 2 identical tables with right angle edges
9. Pennies (250)
10. Kitchen scale
First, make the cup to hold the pennies as follows:
1. Use the 1-hole puncher to make two holes on opposite sides of the cup, near the top edge of the plastic cup.
2. Cut 24 inches of curling ribbon.
3. Tie each end of the ribbon to each of the holes on the cup.
This cup will be used to measure the strength of the structures by hanging it over the structure and measuring the amount of weight it can hold.
Next, make the models representing the nanostructures as follows:
1. Fold six origami papers into a cube.
2. Create six rolls from six origami papers. Roll one at a time. Roll them so each will have two layers of paper all the way around and make sure the edges line up. With squares of 15x15 cm, the diameter of each roll should be 2.5 cm. Secure each roll with a small piece of tape.
3. Stack six origami papers on top of each other.
Now you have made 3 different structures, each from 6 pieces of origami paper. They represent 3 kinds of nanomolecules: densely packed carbon like in diamond (cube), carbon rolled into a hollow structure like fullerenes in a nanotube (rolls), and single sheet graphene layers like the ones found in graphite (stack).
Finally, take the measurements as follows:
1. Place the two identical tables parallel to each other, leaving a gap of about 1 1/2 inches between them. Make sure the gap is small enough so that your cube will not fall through.
2. Start with the stacked sheets structure.
3. Place the structure over the gap with the cup hanging from it, down into the gap. An equal amount of the structure should be resting on each table.
4. Test if the structure can hold the empty cup. Record your findings. Move to step 7 if your structure cannot hold the empty cup. If you see a crack or fold somewhere in the structure that indicates the structure cannot carry the weight.
5. Add weight until the structure cracks, as follows:
a. Hold the cup so the weight of it is in your hands, but so the structure remains in place. Place five pennies in the cup and gently bring the cup back down and let go of it.
b. Note if the structure can hold the extra weight.
c. Repeat steps 5a. and 5b. until your structure shows a crack under the weight.
d. Using the kitchen scale, weigh how much the structure can carry before showing any cracks:
i. Remove the cup with pennies from the structure. Then take out five pennies (since the structure could not hold the weight with the last five pennies added, but it could before you added them).
ii. Measure the weight of the cup with pennies.
iii. Record the measurement in grams.
6. Remove the structure and empty the cup.
7. Repeat steps 3–6 using the six rolls as the structure, placed parallel to each other.
8. Repeat steps 3–6 using the cube as the structure.
9. This completes one trial. Repeat all the steps two more times for a total of three trials (three for each structure). Before you start trial 2, remake the structures. After a structure has cracked once, it will be permanently weakened, so it cannot be used it again for future tests. Make new structures for each new trial.

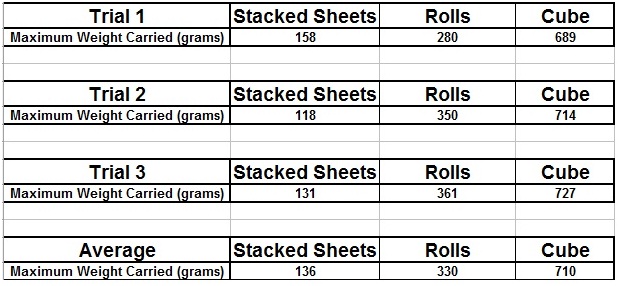
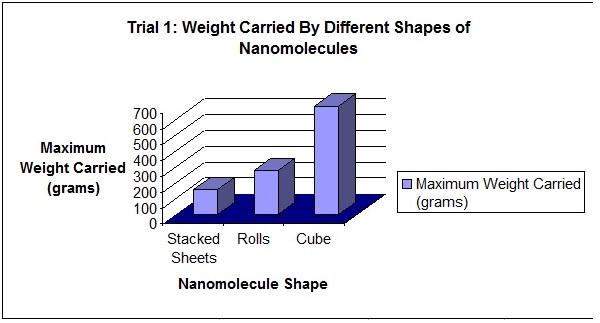
The carbon nanostructure that is the strongest is the cube, the model of the carbon structure representing the diamond. My hypothesis was correct. In each of the three trials, the cube (diamond) held the most maximum weight, a total of 689 grams in the first trial, 714 grams in the second trial, and 727 grams in the third trial. The diamond is the densest of the carbon nanostructures, making it strong but heavy and inflexible. The second strongest nanostructure was the hollow tube, the model of the carbon structure representing the nanotube, a form of a fullerene. Although the nanotube is hollow, it amazingly held a maximum weight of 280 grams in the first trial, 350 grams in the second trial, and 361 grams in the third trial. This is a very good result considering that the structure is hollow and lightweight. The weakest nanostructure was the stacked sheets, the model of the carbon structure representing the graphite. This carbon nanostructure held a maximum weight of 158 grams in the first trial, 118 grams in the second trial, and 131 grams in the third trial.
These nanotubes are the strongest fibers known. When tested in the laboratory, they were found to be 10 to 100 times stronger than steel per unit weight. What makes them amazingly more useful than the extremely strong diamond structure is since they are hollow, they are lightweight. Also, depending on how the nanotubes are rolled up, some forms conduct electricity better than copper wires. What makes nanotubes even more interesting is that they are flexible. These incredible properties have changed the world of science forever. Scientists are researching the use of carbon nanotubes to make artificial limbs, artificial muscles, shape-changing structures, ultra-strong robots, and highly efficient solar cells. Right now, carbon nanotubes are being used to make super-strong, lightweight items such as tennis rackets and bike frames.
They are also being used in prototypes of airplanes, bulletproof clothing, transistors, and ropes that might one day be used to carry an elevator up to space. Also, some other amazing inventions that have been made possible by nanotechnology are water-purifying tea bags that cost just half a cent which would give people access to clean drinking water, nanobots that can diagnose and heal the body from the inside, self-heating roads filled with carbon nano-fibers, fuel cells coated in nanoparticles that can turn human waste into clean, green electricity, and fabric treated with nanorods that make clothes that work as air-filters to clean the air we breathe.
For further experiments with the nanostructure models, it would be interesting to redo the experiment with some additional nanostructures like the buckyball, which is a nanostructure shaped like a soccer ball, and an icosahedron, which is a structure with 20 triangular faces. It would be interesting to find out how strong these nanostructures would be. Also, another experiment could be done to factor in the density of the nanostructure. For example, scientists have found that nanotubes are stronger than steel per unit weight. Steel is much denser and heavier than nanotubes would be and not very useful to make lightweight, strong materials like tennis rackets. Another factor that can be taken into consideration is the distance between the edges of the tables.
As the edges of the tables move closer together, the amount of weight that the nanostructure can hold may change. Also, in order to keep my origami cubes together, I had to tape the edges together. That may have affected their strength. In science this is called a boundary condition. The stacked paper model and tubes may have been able to hold more weight if I had taped their edges together too. This would have changed their boundary condition. For example, taping the edges of the flat sheets together might have kept them from sliding around and made them stronger.
In the nanoworld, boundary conditions have to do with how the different carbon structures bond together. Although my hypothesis was correct and the nanostructure representing the diamond was the strongest, my experiment was based on models I created with origami paper. They were just representing carbon nanostructures to model what happens in the nanoworld. But, it is important to remember that they do not behave exactly like the actual nanoscale structures. Forces do not act the same way in the nanoscale as they do in our everyday scale. Nanotechnology opens up a whole new world for research into the nanoscale that would have to take place in the laboratory.
Conley, Ken. "Episode 72: Underwater Car and Nanotechnology: The Smaller The Stronger." Annotated Mythbusters. Ken Conley, 24 Jan. 2007. Web. 15 Apr. 2014. <http://kwc.org/mythbusters/2007/01/episode_72_underwater_car_and.html>.
Historical Society of Pomona Society. "Folding Paper in Half Twelve Times."Folding Paper in Half Twelve Times. Historical Society of Pomona Society, n.d. Web. 12 Apr. 2014. <http://pomonahistorical.org/12times.htm>.
Stier, Caitlin. "Students Break Record by Folding Toilet Paper 13 times." New Scientist TV. New Scientist, 11 Jan. 2012. Web. 12 Apr. 2014. <http://www.newscientist.com/blogs/nstv/2012/01/paper-folding-limits-pushed.html>.
Weisstein, Eric W. "Folding." From MathWorld-A Wolfram Web Resource, n.d. Web. 18 Apr.2014 <http://mathworld.wolfram.com/Folding.html>